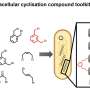
Researchers at the University of Bath have pioneered a groundbreaking technology that utilizes bacteria to construct, chemically stabilize, and evaluate millions of potential drug molecules within living cells. This innovative approach significantly accelerates the process of discovering new treatments for challenging cancer types, which often resist conventional therapies.
The development leverages a method known as peptide stapling, which allows for the creation of highly stable peptides. These peptides can be designed to bind specifically to cancer cells, enhancing the efficacy of targeted therapies. The ability to generate and test vast numbers of these molecules rapidly could lead to breakthroughs in treating cancers that currently have limited treatment options.
Streamlining Drug Discovery
Typically, the process of drug discovery is lengthy and resource-intensive, often taking years before viable treatments can reach clinical trials. By employing bacterial systems to produce these molecules, the researchers have created a much more efficient pipeline.
Dr. Sarah Johnson, lead researcher at the University, stated, “This method allows us to rapidly test the effectiveness of different drug candidates and find those that are most promising. It could change the landscape of cancer treatment.” The bacteria used in this process can swiftly produce thousands of unique peptides, which are then screened for their ability to target specific cancer cells.
The implications of this research are profound. With cancer being one of the leading causes of death globally, advancements in treatment options are crucial. According to the World Health Organization, the global cancer burden is expected to rise to 29.5 million cases by 2040. As traditional therapies often fall short, especially in late-stage cancers, this new technology could provide a much-needed lifeline.
Future Prospects and Collaborations
The research team at the University of Bath is looking to collaborate with pharmaceutical companies to further validate and develop these peptides into effective drugs. The potential for this technology extends beyond cancer, as it could also be adapted for other diseases where targeted treatment is essential.
This innovative approach underscores the importance of interdisciplinary collaboration in tackling complex health issues. The integration of microbiology, chemistry, and oncology exemplifies how new technologies can provide solutions to old problems.
As the project progresses, the team remains focused on ensuring that their findings translate into real-world applications. They are currently seeking funding to expand their research and move towards clinical testing.
In conclusion, the work being done at the University of Bath represents a significant step forward in the quest for more effective cancer treatments. With ongoing advancements in bacterial technology and peptide chemistry, the future of oncology may be poised for transformative change.