A recent study from the University of Barcelona has identified a crucial molecular mechanism that explains how cells communicate through extracellular vesicles (EVs). Published on November 28, 2025, in the Journal of Extracellular Vesicles, this research reveals the role of the Commander protein complex in coordinating the entry and internal routing of these vesicles within cells.
Led by Professor Albert Lu from the Faculty of Medicine and Health Sciences and the CELLEX Biomedical Research Center (IDIBAPS-UB), the study highlights the Commander complex’s involvement in the recycling of cellular membranes. This discovery is vital for understanding intercellular communication, which is essential for developing new therapies and diagnostic tools.
Professor Lu emphasized the significance of this research, stating, “Understanding how receptor cells capture and process extracellular vesicles is essential to understanding how our body communicates at the molecular level.” He further noted that this knowledge could enhance the therapeutic and diagnostic potential of EVs by ensuring they reach the appropriate target cells.
Innovative Methodology Using CRISPR-Cas9
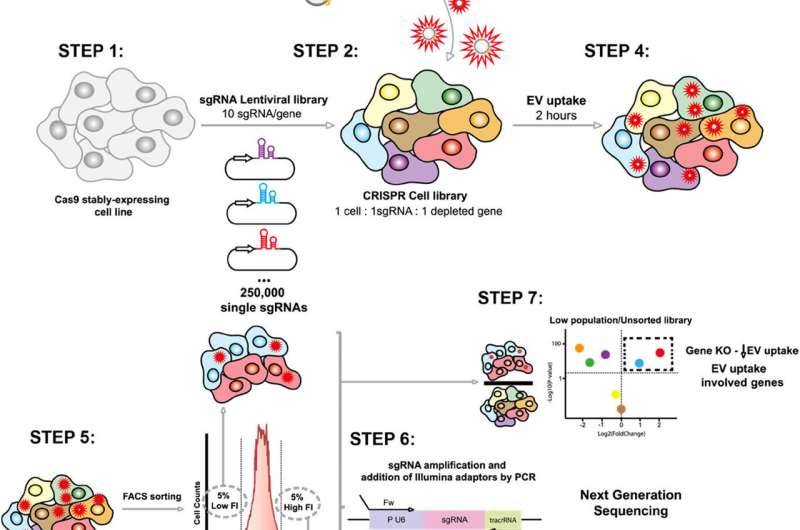
Extracellular vesicles are small particles secreted by all cells, acting as biological messengers that transport proteins, lipids, and nucleic acids. To investigate the mechanisms that govern their uptake, the researchers employed a groundbreaking approach. They utilized CRISPR-Cas9 technology to conduct a massive genomic screening, systematically deactivating each of the more than 20,000 human genes.
Each group of genetically modified cells had a specific gene disabled. The cells were then exposed to EVs marked with a fluorescent dye, allowing the researchers to measure which cells captured more or fewer vesicles using flow cytometry. Following this, fluorescence-activated cell sorting (FACS) was employed to isolate cells with varying uptake capabilities. The deactivated genes were identified through mass sequencing.
“This systematic and unbiased approach allows us to discover new regulators without relying on prior hypotheses, unlike traditional techniques,” explained Professor Lu.
The study found that the Commander endosomal recycling complex, composed of multiple proteins, serves as a fundamental regulator in vesicle uptake. The research, conducted across various human cell lines, suggests that this mechanism is conserved and potentially universal, though its activity may vary depending on the specific cell type or physiological context.
Implications for Therapeutic Development
The findings hold significant therapeutic promise. The ability of EVs to traverse cellular membranes and target specific tissues positions them as natural vehicles for drug delivery or therapeutic molecules. “Understanding how their entry, intracellular trafficking, and delivery of molecular cargo are regulated opens the door to designing EVs with controlled directionality,” Professor Lu noted. This could enhance their efficacy in regenerative, oncological, or anti-inflammatory therapies.
Ongoing research aims to further explore the Commander complex’s role in regulating vesicle uptake and to determine whether this mechanism operates in other cell types or tissues. The researchers are also investigating whether alterations in the Commander complex contribute to disrupted cell communication in pathological conditions, such as cancer or neurodegenerative diseases.
“The long-term goal is to manipulate this pathway to modulate communication between cells and improve the use of EVs as therapeutic and diagnostic tools,” concluded Professor Lu.
This study underscores the potential of extravesicular communication in advancing medical science, paving the way for innovative therapies that exploit the natural pathways of cellular interaction.