Research from the University of Auckland has uncovered critical insights into the evolutionary origins of bacterial motors, which emerged approximately 3.5 billion to 4 billion years ago. This study provides a detailed understanding of bacterial stators, proteins that function similarly to pistons in car engines and are essential for the mobility of bacteria.
Dr. Caroline Puente-Lelievre, a researcher in the School of Biological Sciences, explains that stator proteins are embedded within the bacterial cell wall. They convert charged particles (ions) into torque, enabling bacteria to swim through liquid environments. This research, published in the journal mBio, suggests that stators likely evolved from ion transporter proteins, which are prevalent in bacterial cell membranes.
Unveiling Ancient Mechanisms of Motion
The research was conducted in collaboration with UNSW Sydney and the University of Wisconsin Madison. A significant breakthrough came from DeepMind AI’s AlphaFold technology, which predicts the three-dimensional structures of proteins, facilitating the exploration of these ancient biological systems. Bacteria, among Earth’s earliest life forms, surfaced in a time when the planet was volatile, characterized by intense volcanic activity and meteorite bombardment.
In this demanding environment, bacteria evolved as single-celled organisms equipped with advanced internal motors. Stators generate the necessary power to turn rotors, which in turn spin the flagellum—a long tail that propels the cell, akin to a microscopic propeller. The Latin term “flagellum” translates to whip, aptly describing the tail’s movement.
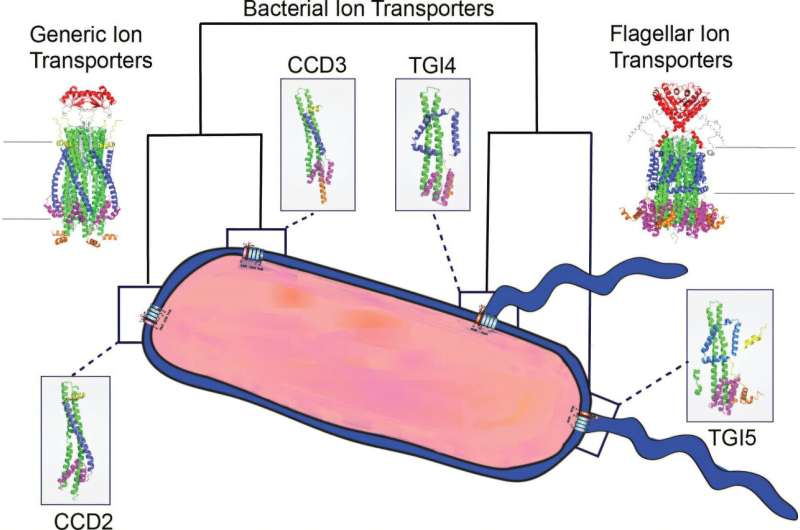
To further investigate stators, researchers analyzed genomic data from over 200 bacterial genomes. They constructed evolutionary trees, utilized advanced computational tools, modeled 3D protein structures, and conducted laboratory experiments. Understanding the 3D shape of these proteins was essential, as shape is integral to function.
Tracing the Evolution of Stator Proteins
Dr. Puente-Lelievre noted, “We predicted the sequences and structures of ancestral proteins that existed millions or billions of years ago and may no longer exist.” Each stator typically comprises five identical versions of a protein named MotA and two identical versions of a protein called MotB. These motor proteins are thought to have evolved from an ancient two-protein system, which later adapted to serve various functions, according to Dr. Nick Matzke, a senior researcher at the University of Auckland.
“This supports the idea that complex machines evolve by coopting simpler machines with simpler functions,” Matzke explained. Just as the ancestors of modern birds likely adapted protofeathers for flight, ancient bacteria repurposed ion flow mechanisms, transforming them into enduring engines of movement.
The research team compared 3D protein structures to identify key differences among similar proteins and the unique traits of stators, particularly regions responsible for generating torque. “We took E. coli bacteria that lacked the torque-generating interface and found that none of them could swim,” Puente-Lelievre confirmed. This result underscores the importance of the specific region for locomotion in this group of bacteria.
Despite billions of years of evolutionary change, the fundamental characteristics of these microscopic engines have remained strikingly consistent. Assistant Professor Matthew Baker from UNSW Sydney remarked on the current state of structural biology and microbiology, stating, “We live in a remarkable era where new sequences are discovered daily, and tools like AlphaFold allow us to quickly explore possible protein structures.”
This study casts a wide net across various species, illuminating the shared and distinct features of stator-like proteins while uncovering the historical evolution of these biological motors. The findings not only enhance our understanding of bacterial motility but also provide a glimpse into the intricate machinery that supports life on Earth.