AI biology company Prima Mente has introduced a groundbreaking approach to diagnosing early-stage Alzheimer’s disease by leveraging advancements in epigenomic analysis. This innovative model aims to address significant challenges in understanding neurodegenerative conditions, which often manifest subtle changes long before symptoms are evident.
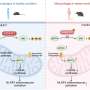
The company’s AI platform, known as Pleiades, utilizes DNA methylation changes found in cell-free DNA (cfDNA) extracted from blood samples. This technique offers a less invasive and more cost-effective alternative to traditional brain biopsies, which are currently the standard for diagnosing such conditions. Remarkably, Pleiades achieved an 89% accuracy rate in detecting early-stage Alzheimer’s and Parkinson’s diseases in real-world clinical trials. When combined with leading neurodegenerative protein biomarkers like pTau-217, the accuracy skyrocketed to 97%.
Advancements in Epigenomics and AI Collaboration
Founded in 2022, Prima Mente is headquartered in London, with additional offices in San Francisco and Dubai. The company’s mission is to redefine precision diagnostics by focusing on the dynamic nature of the epigenome, which plays a crucial role in aging biology. Co-founder Hannah Madan, PhD, emphasized the importance of their approach, stating, “Imaging and cognitive scores don’t necessarily tell you how to intervene or change the path of disease. This is what epigenetics can start to unravel.”
To develop Pleiades, Prima Mente has partnered with NVIDIA and Nebius, utilizing the DGX Cloud Lepton AI platform. This collaboration allows the team to harness advanced computational resources and expertise necessary for building complex models that can understand biological processes over time. The platform is currently being used in a live clinical study named SANDBOX, which spans 15 sites across the U.K. The goal is to enhance classification and stratification of patients suspected of neurodegenerative diseases, thereby improving clinical outcomes and assisting the U.K. National Health Service in managing waiting lists.
Future Directions and Therapeutic Innovations
Pleiades is built on a foundation of 7 billion parameters, trained using 1.9 trillion tokens of epigenetic data that reflect human methylation and genomic sequences. The model’s ability to pinpoint the cfDNA cell type of origin is a significant advancement, as it identifies brain components, such as neurons and microglia, as well as immune cells, which are crucial predictors of Alzheimer’s disease.
Additionally, Pleiades has revealed that cfDNA fragmentation patterns can serve as a new class of biomarkers for detecting Alzheimer’s. These fragments provide insights into various biological processes, including epigenetic states and mechanisms of cell death. With an impressive 83% per-nucleotide accuracy in reconstructing cfDNA fragments, the model enhances signals from specific blood cell types by up to 20-fold, facilitating the detection of rare disease signatures from limited samples.
Looking ahead, Madan expressed the company’s aspirations to expand the capabilities of Pleiades beyond epigenomics to include proteomics, transcriptomics, and additional clinical data. Current longitudinal studies are underway, incorporating both blood samples and brain tissue to further validate this approach. By 2026, Prima Mente plans to apply its generalizable platform across various disease areas, marking a significant step forward in the quest for effective diagnostics and therapies in neurodegenerative diseases.