A research team led by Dr. Sung Mook Choi from the Korea Institute of Materials Science (KIMS) has developed a groundbreaking non-precious metal catalyst for oxygen evolution reactions, aimed at enhancing hydrogen production efficiency. Collaborating with a team led by Professor Seung-Hwa Lee at Changwon National University, the researchers designed a layered catalyst structure optimized for anion exchange membrane water electrolysis (AEMWE), setting a new benchmark in hydrogen production technology.
This innovative catalyst offers a solution to the high costs associated with precious metal catalysts traditionally used in hydrogen production. The significance of this research lies in its ability to deliver both high efficiency and durability while employing cost-effective materials. AEMWE operates under alkaline conditions, allowing for the use of non-precious metal catalysts, which can significantly reduce production costs and enhance safety.
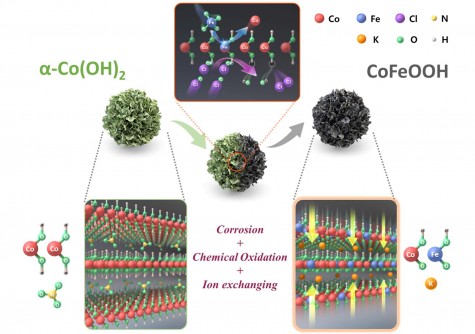
One of the major obstacles in AEMWE technology has been the lack of durable non-precious metal oxygen evolution reaction (OER) catalysts. Conventional transition metal-based catalysts have often encountered issues such as structural degradation and metal dissolution during prolonged operations. To combat these challenges, the research team engineered a cobalt (Co) and iron (Fe)-based oxyhydroxide (CoFeOOH) catalyst with a carefully designed layered structure. This design allows for a more stable catalytic surface and improved charge transfer efficiency during the oxygen evolution reaction.
To further enhance the catalyst’s performance, the researchers incorporated iron into the CoFeOOH structure. This integration modulates the electronic states of the cobalt centers, effectively lowering the energy barriers for crucial reaction steps. Consequently, the catalyst achieved impressive current densities at low overpotentials and maintained stable performance over extended durations without degradation.
Advancing Towards Commercialization
In addition to optimizing the catalyst’s structure, the team developed a proprietary technique for controlled chemical oxidation of the catalyst surface. This method helps prevent corrosion and structural degradation commonly associated with iron doping. The result is a robust catalyst that performs reliably under alkaline conditions, making it suitable for practical applications.
The newly developed catalyst was tested in a unit cell of AEMWE, demonstrating both performance and durability under real-world electrolysis conditions. This validation marks a significant step toward the practical implementation of non-precious metal OER catalysts in AEMWE systems.
Once commercialized, this technology is expected to facilitate the development of affordable, efficient AEMWE systems, reducing reliance on precious metals. Moreover, it holds promise for advancing clean hydrogen production and enhancing technological self-sufficiency in key materials for water electrolysis catalysts.
Dr. Sung Mook Choi, Principal Researcher at KIMS, remarked, “This research represents a case in which the limitations of non-precious metal–based catalysts were overcome through structural design.” He emphasized the team’s commitment to accelerating the commercialization of green hydrogen technologies, contributing to a future-oriented hydrogen-based society.
The findings of this research were published online on December 1, 2025, in the prestigious journal ACS Nano, which has an impact factor of 16.1. The study received support from the National Research Laboratory for Hydrogen (H2 NEXT ROUND) program of the National Research Foundation of Korea (NRF), as well as various institutional research programs.
KIMS, as a non-profit government-funded institute under the Ministry of Science and ICT of the Republic of Korea, plays a crucial role in advancing materials science and technology. By focusing on comprehensive research and development, KIMS continues to contribute significantly to the growth of the Korean industry and the global materials science landscape.