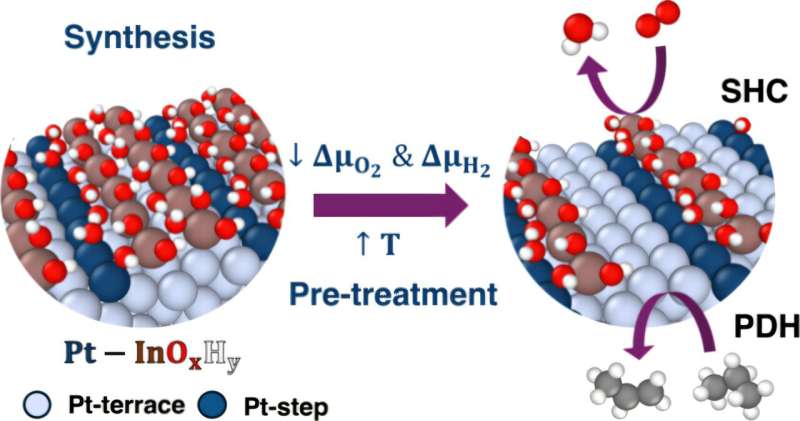
Researchers at the University of Rochester have developed advanced algorithms that enhance the understanding of how propane is transformed into propylene, a crucial component in numerous everyday products. This breakthrough, detailed in a study published on November 13, 2025, in the Journal of the American Chemical Society, reveals key atomic features that drive this complex chemical process.
Propylene, used in items ranging from plastic squeeze bottles to outdoor furniture, is produced by converting propane through a method that can now be optimized thanks to new insights from these algorithms. Previous research published in 2021 demonstrated that chemists could use tandem nanoscale catalysts to streamline this conversion process, increasing yield while reducing costs. However, the underlying atomic mechanisms remained largely unclear, posing challenges for broader industrial applications.
Understanding Atomic Interactions
Siddharth Deshpande, an assistant professor in the Department of Chemical and Sustainability Engineering, emphasized the complexity of the catalytic active sites involved in this reaction. “There are so many different possibilities of what’s happening at the catalytic active sites, so we need an algorithmic approach to very easily yet logically screen through the large amount of possibilities that exist and focus on the most important ones,” he explained.
Deshpande, along with Ph.D. student Snehitha Srirangam, utilized refined algorithms to conduct a detailed analysis of both the metallic and oxide phases that influence this intricate reaction. Their findings revealed unexpected results regarding the behavior of oxides in the chemical reaction, particularly their preference for developing around defective metal sites. This specific interaction proved to be critical for maintaining catalyst stability.
Despite the varied chemical environments in which oxides can exist, the researchers observed that their functional role remained consistent. This knowledge could pave the way for enhanced efficiencies in the production of propylene and other critical industrial materials.
Implications for Industrial Chemistry
The implications of this research extend beyond propane conversion. Deshpande suggests that the algorithmic approach could also be applied to other chemical reactions, such as methanol synthesis, which is essential for products like paints and fuel cells. “Our approach is very general and can open the doors to understanding many of these processes that have remained an enigma for decades,” he remarked.
By leveraging this new understanding, companies may be able to reduce reliance on traditional trial-and-error methods that have dominated the industry for years. Deshpande asserts that, while the mechanisms for producing these chemicals are well-established, comprehending the “why” behind their effectiveness remains a crucial area for further exploration.
As the field of chemical engineering continues to evolve, the insights gained from this study may lead to more strategic and efficient methods for producing essential materials, potentially transforming multiple industries reliant on chemical processes.
For more information, refer to the study by Snehitha Srirangam et al., titled “Site-Selective Oxide Rearrangement in a Tandem Metal–Metal Oxide Catalyst Improves Selectivity in Oxidative Dehydrogenation of Propane,” published in the Journal of the American Chemical Society. The DOI for the study is 10.1021/jacs.5c13571.